sop for Electronic Data Backup
1.0 PURPOSE
To define a procedure for electronic analytical and any other computerized system data backup.
2.0 SCOPE
2.1 This procedure shall be applicable for the backup/restore of all electronic analytical data
2.1.1 This guideline shall be applicable for analytical data backup for Example: HPLC/GC/UV/AAS and Auto- titrator.
2.1.2 This guideline shall be applicable for any other computerized system data. For example BMS and SCADA data backup.
3.1 References
In-House
3.2 Attachments
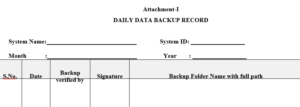
3.2.1 Attachment-I : Daily backup record
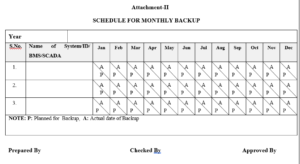
3.2.2 Attachment-II : Schedule for Monthly backup
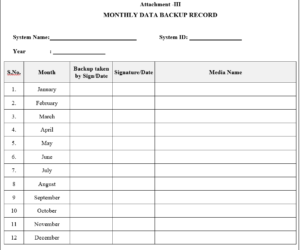
3.2.3 Attachment-III : Monthly data backup record
3.2.4 Attachment-IV : Data verification record for HPLC/ GC.
3.2.5 Attachment–V : Data verification record UV/AAS/Autotitrator.
3.2.6 Attachment–VI : Data verification record other than analytical data (e.g. BMS/SCADA).
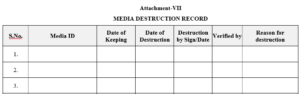
3.2.7 Attachment-VII : Media destruction record.
4.0 DEFINITION
4.1.1 Backup: Backup is the process of copying active files from online disk to external media so that files may be restored to a disk in the event of equipment failure, damage to or loss of data.
4.1.2 Computerized system: A system including the input of data, electronic processing and the output of information to be used either for reporting or automatic control.
4.1.3 Restore: Restore the process of bringing off line storage data back from the offline media and putting it on an online storage system such as a file server.
4.2 Abbreviations
4.2.1 AAS : Atomic Absorption Spectroscopy
4.2.2 BMS : Building Management System.
4.2.3 CPU : Central Processing Unit
4.2.4 CD : Compact Disc
4.2.5 DVD : Digital versatile Disc
4.2.6 GC : Gas Chromatography
4.2.7 HPLC : High Performance Liquid Chromatography.
4.2.8 SCADA : Supervisory Control And Data Acquisition
4.2.9 IT : Information technology
4.2.10 UV : Ultra-violet
5.0 RESPONSIBILITY
5.1 User department
5.1.1 To take backup of the electronic data.
5.1.2 To ensure timely backup data
5.1.3 To maintain all backup records.
5.2 Site IT department:
5.2.1 To help in taking backup of the electronic data.
5.2.2 To co-ordinate and help for backup.
5.3 QC/User department Head:
5.3.1 To verify the data backup record and files.
5.3.2 To identify the required data for backup.
5.3.3 To ensure daily and monthly backup.
5.3.4 To ensure data restoration and verification of data.
5.4 Quality Assurance Head:
5.4.1 To ensure implementation of defined procedure as per SOP.
5.5.2 To provide the necessary support regarding data backup.
5.5 Plant Head:
5.5.1 To ensure implementation of defined procedure as per SOP.
5.5.2 To provide the necessary support regarding data backup.
6.0 Distribution:
I. Quality Assurance Department
II. Quality Control Department
III. Production Department
IV. Ware house Department
V. Engineering Department
7.0 PROCEDURE:
7.1 Daily backup:
7.1.1 User shall create folder daily on system administrator path.
7.1.1.1 Daily backup shall be taken by system administrator with year, month & date wise folder.
7.1.1.2 QC/user department head shall ensure that daily backup is taken on correct path by system administrator.
7.1.1.3 Daily data backup shall be taken in hard disk of system.
7.1.1.4 Daily data backup record shall be maintained as per Attachment –I with full path of file. For example:
D/system/year/month/date
7.1.1.5 All QC critical instruments (used for quantitative analysis) and which are connected to CPU shall be covered for daily backup. For e.g. HPLC / UV/ GC/ AAS / Auto-titrator etc.
7.1.1.6 Any other computer system, data shall be covered for daily backup. For e.g. BMS/SCADA system.
7.1.1.7 Daily date wise analytical data/any other computer system data shall be in particular year, month folder and may be deleted from hard disk after monthly backup.
7.1.2 Monthly backup
7.1.2.1 QC/user department deputed person shall prepare the schedule of monthly backup as per Attachment –II for all critical instruments (used for quantitative analysis), as per # 7.1.1.5and any other computer system data as per # 7.1.1.6
7.1.2.2 Monthly backup shall be taken at end of calendar month as per Attachment–II (schedule for monthly backup) by the concern department with the help of IT person.
7.1.2.3 QC/user department head shall be responsible for Monthly backup & shall take backup on monthly basis.
7.1.2.4 Monthly data backup shall be taken in CD/DVD/ Blu ray disk or any external drive.
7.1.2.5 Monthly data backup record shall be maintained as per Attachment –III.
7.1.2.6 If, there is any version change of software, old version shall be archived for future reference, if practicable.
7.1.3 Verification of data of backup process.
7.1.3.1 QC/user department person shall select single product analytical data/any computer system data from laboratory instrument/BMS/SCADA through computers running the application software for processing and print the data at the time of monthly backup.
7.1.3.2 QC/user department person shall take processed data print on paper.
7.1.3.3 The printed data shall be reviewed by the QC Head/user department head with signature on data.
7.1.3.4 Printed reviewed data in the paper form with original signature shall be treated as master copy and same shall be used for taking all decisions pertaining to quality of the data.
7.1.3.5 Reviewed data shall be verified on quarterly basis till the media storage time by the QC person/user department person from the backup, restores data.
7.1.3.6 Similarly, a product analytical data/any other computer system data shall be selected for checking every month and that data shall be checked by QC person/user department, quarterly by the QC person/user department from the backup data by restoring the desired data.
7.1.3.7 QC/user department person shall check data backup for any variation and keep as record.
7.1.3.8 Verification record shall be maintained as per Attachment- IV/V/VI.
7.1.3.9 Separate record shall be prepared for each instrument.
Note: For any other instrument/computer system, verification records can be prepared as per attachment VI.
7.1.4 Restoration of backup data
7.1.4.1 QC/User concern person shall restore the desired data in desired system.
7.1.4.2 QC/User department head shall ensure correct data restore in desired system.
7.1.4.3 QC/User department head shall check and verify the restore data at defined interval.
7.1.5 Archive and Storage condition
7.1.5.1 Daily backup media shall be archived in hard disk of system administrator.
7.1.5.2 Monthly backup containing media shall be archived at an approved local site or outside storage facility in lock and key under triple layer security.
7.1.5.3 Both the local site storage and outside storage location, if used shall meet the following criteria regarding storage condition.
7.1.5.3.1 Only authorized person accessibility.
7.1.5.3.2 Adequate environmental controls (e.g. temperature 25 +/-2*C, humidity 50 +/- 5 %) or any specific condition specified by media manufacturer.
7.1.5.3.3 Proper media storage space.
7.1.6 Media identification and availability.
7.1.6.1 Media labeling shall be labeled by QC person / user department.
7.1.6.2 For monthly backup media shall be labeled as backup_system name_MMYYYY with date and signature.
7.1.7 Removal of media from archive
7.1.7.1 Media shall be removed from archive only as required for restoration, testing the restoration, destruction, whenever required for verification of backup data and recovery procedures.
7.1.7.2 Only authorized person shall be permitted to remove media from the storage facility.
7.1.7.3 Shelf life for media shall be for seven years from the date of backup and after that media shall be destroyed by the authorized person as per Attachment -VII.
7.1.8 Media destruction
7.1.8.1 Media shall be destroyed by the shredders method by reducing the media into small pieces at least three pieces.
7.1.8.2 Grinding and hammer-milling process can use for destruction of media to reduce solid material to tiny fragments.
7.1.8.3 Destruction shall be in presence of only authorized person with authorization details and shall be recorded as per Attachment -VII.