sop for monitoring of water purification and distribution system
1.0. OBJECTIVE:
The objective of this SOP is:
1.1 To describe the procedure for monitoring of Water Purification, Storage and Distribution System in Cephalosporin Block and Warehouse.
2.0. RESPONSIBILITY:
2.1 The QC Officer shall be:
2.1.1. Responsible for sampling as per the sampling schedule.
2.1.2. Responsible for Analysis of samples as per the respective SOPs.
2.1.3. Responsible for reporting of results of different types of water in respective formats.
2.2 The QC – Executive shall be:
2.2.1 Responsible for ensuring the adherence of the SOP.
3.0. ACCOUNTABILITY:
Head – Quality Control.
4.0. PROCEDURE:
4.1 Sampling Points:
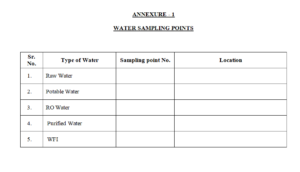
4.1.1 Water monitoring shall be carried out from the various storage tanks and user points as specified in the sampling points. (Refer Annexure – 1). This annexure specifies all the points to be covered under monitoring plan.
4.2 Sampling Frequency:
4.2.1 The frequency of monitoring of water quality from final storage tanks of Purified Water and Water For Injection is daily and that for users points are on rotation basis, (every points is covered for analysis at least once in a week.
4.2.2 Refer Annexure – 2 for sampling points due for monitoring for the day.
4.3 Specification:
4.3.1 Water from all the final storage tanks of Purified Water and Water For Injection shall be subjected for complete analysis, including Chemical and Microbiological Test as per following specification numbers.
| Sr. No. | Water Grade | Specification No. |
|
|
Raw Water | |
|
|
Potable Water | |
|
|
Reverse Osmosis Water | |
|
|
Purified Water | |
|
|
Water For Injection |
4.4 Sampling Procedure:
4.4.1 Sample shall be collected from the sampling points due on a particular day, separately for chemical and Microbiological analysis.
4.4.2 For sampling preparation and sampling procedure refer SOP
4.5 Analysis:
4.5.1 Analysis of Water is grouped under two sections i.e. “X” & “Y”.
4.5.2 “X” is complete analysis and “Y” is description, pH and Microbiological Analysis. The extent of analysis is referred in front of the sampling schedule in Annexure – 2
4.5.3 Analysis of Purified water and water for injection shall be carried out as per British Pharmacopoeia.
Rationale:
Water purification system has been subjected to Performance of system for last four months, where it was found that the chemical purity at Point of Use (POU) remains same as that of Storage Tanks.
The distribution of Purified Water / Water For Injection from Storage tank to user point is done through enclosed circulation loop, which is made of SS316L. Thus the chemical purity of water remains unaltered.
4.6 Reporting:
4.6.1 Analysis records for Chemical analysis and Microbiological analysis shall be prepared for different types of water as per the respective formats given in Annexure – 3.
4.6.2 Trend chart for Microbial Count shall be prepared for the data observed for one month.
4.6.3 Certificate of Analysis shall be prepared for Purified water and Water for injection from the storage tank in as per the format given in Annexure – 4 and 5.
5.0. REASON FOR REVISION:
– Sampling point and sampling frequency has been revised based on performance of the system for last four months.
– Specification of water of different grades has been revised.
– Reporting logbook has been designed.
6.0 TRAINING:
Trainer — Head – Quality Control
Trainees — Quality Control Executives / Officers
Period — One day
7.0 DISTRIBUTION:
Certified Copy No. 1 : Head of Department – Quality Control
Certified Copy No. 2 : Head of Department – Engineering
Certified Copy No. 3 : Head – Plant Operations
Original Copy : Head – quality assurance.
8.0 ANNEXURES:
Annexure – 1 : Water Sampling Points
Annexure – 2 : Sampling Schedule
Annexure – 3 : Formats for Analysis Record
Annexure – 4 : Format for Certificate of Analysis of Purified Water
Annexure – 5 : Format for Certificate of Analysis of Water For Injection
9.0 REFERENCES:
In house