analysis of silodosin 8 mg capsule
TEST PROCEDURE / METHODS
standard testing procedure of silodosin 8 mg capsule
01. Description: Remove 20 Capsules from the strip. Place on a white paper, open the capsule and observe visually.
02. Average filled Weight : Weigh accurately 20 Capsules and Open the capsule without losing any part of the shell and remove the contents as completely as possible. Weigh the shell. The weight of the contents is the difference between the weighings.
Average filled Weight = Weight of 20 Capsules – Weight of 20 empty Capsules / 20
03. Uniformity of filled Weight Put one Capsule at the pan of Analytical balance tare the balance & pick up the capsule, open it withdraw filled powder and clean both body & cap of capsule again put the empty capsule note the weight of the balance display i.e. the weight of the filled content, repeat the same operation for other nineteen capsules to find the Uniformity of the filled weight. & record. Not more than two of the individual weights deviate from the average weight by more than 10% and none deviates by more than 20%.
04. Identification
In the assay, the principle peak in the chromatogram obtained with the test solution corresponds to the peak in the chromatogram obtained with reference solution.
05 Leak test Equipment:
a) A vacuum Desiccator.
b) A vacuum pump with gauge.
Procedure: Take 3 strips and keep in vacuum desiccator. Ensure that strips are immersed in water. Connect the desiccator to vacuum pump with rubber tube and apply vacuum about 381 mm (15”) of mercury. The vacuum should be maintained for 30 sec. Release vacuum. Remove the strips wipe and dry with a clean cloth cut the strip and examine carefully. None of the Capsules should be sticky or moist.
06 Disintegration Test Place six Capsules one each in six tubes of the basket in a suitable disintegration test apparatus. Previously suspend the assembly in water (about 750-800 ml) maintained at a temperature of 37˚C ± 1˚C and operate the apparatus with disc to each tube. The Capsules pass the test, if all the Capsules have disintegrate (no residue of the Capsules remains on the screen of the apparatus or, if a residue remains, it consists of fragments of insoluble coating of the Capsules or is a soft mass with no palpable core) in 30 minutes. If 1 or 2 Capsules fail to disintegrate, repeat the test on 12 additional Capsules, not less than 16 of the total of 18 Capsules tested disintegrate. If the Capsules adhere to the disc and the preparation being examined fails to comply, repeat the test omitting the disc.
7.0 Uniformity of Content Chromatographic system
– A stainless steel column 25 cm x 4.6 mm packed with Octylsilane bonded to porous silica (5μm)
– Mobile phase: a mixture of 50:50 volumes of 0.1% OPA in water, & methanol.
– flow rate 2 ml per minute,
– spectrophotometer set at 269 nm,
– Injection volume: 20 μl.
Standard Preparation: Weight Accurately 80mg WS of Silodosin in 100ml of VF dissolve in 70ml of Methanol and dilute to 100 ml with Methanol further dilute 10 ml in 50 ml of VF and make up to 50ml with Mobile phase.
Test Preparation: Take 10 Capsules open & add powder separately into 10 50ml of VF sonicate to dissolve in 30ml of Mobile phase and make up to 50ml with Mobile phase.
After saturation of column Separately inject the 20µl of filtered Standard & Test solution. record the chromatogram and measure the peak response with the reference chromatogram
Calculation for Uniformity of Content:
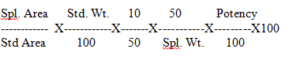
8.0 Assay-
Silodosin-8 mg Estimation of Silodosin By liquid chromatography
Chromatographic system
– A stainless steel column 25 cm x 4.6 mm packed with octylsilane bonded to porous silica (5μm)
– Mobile phase: a mixture of 50:50 volumes of 0.1% OPA in water, & methanol.
– flow rate 2 ml per minute,
– spectrophotometer set at 269 nm,
– injection volume: 20 μl.
Standard Preparation: Weight Accurately 80mg WS of Silodosin in 100ml of VF dissolve in 70ml of Methanol and dilute to 100 ml with Methanol further dilute 10 ml in 50 ml of VF and make up to 50ml with Mobile phase.
Test Preparation: Collect the 20 Capsules powder weight Accurately Eq. to 80 mg of Silodosin in 100ml of VF sonicate to dissolve in 70ml of Methanol and dilute to 100ml with Methanol. further dilute 10 ml in 50 ml of VF and make up to 50ml with Mobile phase
After saturation of column Separately inject the 20µl of Standard & Test solution. record the chromatogram and measure the peak response with the reference chromatogram
Calculation:
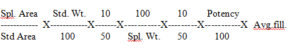
(Calculate the result of labeled amount of Silodosin, Since the labeled amount of Silodosin is 8 mg.
Abbreviations:
OPA :-Orthophosphoric acid
WS :- working standard
VF :-Volumetric flask
STD :-Standard
Spl. :-Sample
| Supersede No | Effective Date | Reason for Revision |
| Nil | NA |
standard testing procedure glass ampoule
Standard testing procedure of Iron Sucrose Injection
Standard testing procedure lactose
Standard testing procedure mefenamic acid
standard testing procedure domperidone
standard testing procedure flavour mixed fruit
standard testing procedure dicyclomine hydrochloride
Standard testing procedure honey pure
standard testing procedure dextromethorphan hydrobromide
standard procedure of levocarnitine injection
Analysis of Ivermectin Suspension
standard testing procedure artemether injection
standard testing procedure artemether injection
standard testing procedure Carbocisteine syrup
standard testing procedure Phytomenadione injection
standard testing procedure serratiopeptidase
standard testing procedure starch IP
standard testing procedure sucrose refined sugar
standard testing procedure titanium dioxide
standard testing procedure tramadol hydrochloride
standard testing procedure zinc sulphate
standard testing procedure croscarmellose sodium
standard testing procedure colour erythrosine supra
standard testing procedure magnesium hydroxide
standard testing procedure diclofenac sodium
standard testing procedure dibasic calcium phosphate
standard testing procedure cyanocobalamin
standard testing procedure cholecalciferol
standard testing procedure Calcium carbonate oyster shell powder
standard test procedure Calcium Citrate
standard testing procedure Bronopol
standard testing procedure Bromhexine Hydrochloride
Standard Testing Procedure diclofenac sodium injection
Standard Testing Procedure Drotaverine Hydrochloride injection