Analysis of Losartan Tablets
TEST PROCEDURE / METHODS
01. Description: Remove 20 tablets from the strip. Place on a white paper and observe visually.
02. Identification
In the assay, the principle peak in the chromatogram obtained with the test solution corresponds to the peak in the chromatogram obtained with reference solution.
03. Average Weight: Weigh accurately 20 tablets and calculate the average weight.
Average Weight = Weight of 20 tablets/20
04 Dissolution Apparatus. No 1, (Paddle)
Medium. 900 ml of water
Speed 50 rpm, and Time-45 minutes.
Withdraw a suitable volume of the medium and filter.
To 10 ml of filtrate and dilute to 25 ml with water.
Standard Preparation
Weigh accurately (about 55 mg) dissolve in 50 ml Water heat upto 40 C, sonicate for 1 minute and dilute to 100 ml with Water,
To 5ml dilute to 100 ml with water and filter.
To 10 ml dilute to 25 ml with water.
Calculation

D. Not less than 75%
Procedure – Measure the absorbance at 250 nm with UV & Visible
Spectrophotometer.
5.0
Related Substances Determine by liquid chromatography
Buffer – Weight (about 770 mg) ammonium acetate dissolve in 900 ml water + 2 ml Triethylamine adjust pH- 6.5 with glacial acetic acid and dilute to 1000 ml with water.
Mobile phase. A mixture of 75V buffer and 25V Acetonitrile.
Column – 250 x 4.0 mm, 5µm (C8)
Flow Rate -1.5 ml per minute
Wavelength – 235 nm
Injection volume- 10 µL
Run Time – 40 minutes
Sequence of Injection
| S.No. | Particular | No. of Injection |
| 1 | Check RT | 1 |
| 2 | Standard | 5 |
| 3 | Test | 2 |
| 4 | Column conditioning | 1 |
| 5 | RS Placebo | 1 |
| 6 | Reference Solution B (RS) | 1 |
| 7 | Test Solution (RS) | 1 |
Test Preparation
Weigh and powder of 20 tablets take powder eq. to 100 mg of Losartan Potassium (about 680 mg) dissolve in 50 ml water and dilute to 100 ml with water.
Reference solution A
Weigh accurately (about 50 mg) Losartan potassium WS dissolve in 40 ml water and dilute to 50 ml with water.
Reference Solution B
To 1 ml of Ref Solution A dilute to 100 ml water.
Column Efficiency
Tailing Factor – Limit NMT NMT 3.0
Theoretical Plates – Limit NLT 1000
Calculation

6.0 Assay By High Performance Liquid Chromatography
Buffer- 770 mg ammonium acetate dissolve in 1000 ml water
Mobile phase- 650 Buffer +300 Acetonitrile +50 ml methanol+2 ml Triethylamine and adjust pH- 6.6 with Glacial acetic acid.
Column – 250 x 4.0 mm, 5µm (C8)
Flow Rate -1.0 ml per minute
Wavelength – 237 nm
Injection volume- 20 µL
Standard Preparation
Weigh accurately (about 65 mg) Losartan Potassium WS dissolve in 20 ml mobile phase heat upto 40°C, sonicate for 1 minute and dilute to 50 ml with mobile phase.
To 5ml dilute to 50 ml with mobile phase.
Test Preparation
Weigh and powder of 20 tablets take powder about (442 mg) mg dissolve in 20 ml mobile phase heat upto 40 C, sonicate for 1 minute and dilute to 50 ml with mobile phase.
To 5ml dilute to 50 ml with mobile phase.
Column Efficiency
Tailing Factor …………….. Limit NMT 2.0
Theoretical Plate ……………… Limit NLT 5000
Calculation
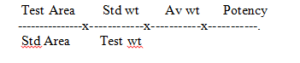
Abbreviations:
WS :- working standard
VF :-Volumetric flask
Std :-Standard
Spl :-Sample
standard testing procedure glass ampoule
Standard testing procedure of Iron Sucrose Injection
Standard testing procedure lactose
Standard testing procedure mefenamic acid
standard testing procedure domperidone
standard testing procedure flavour mixed fruit
standard testing procedure dicyclomine hydrochloride
Standard testing procedure honey pure
standard testing procedure dextromethorphan hydrobromide
standard procedure of levocarnitine injection
Analysis of Ivermectin Suspension
standard testing procedure artemether injection
standard testing procedure artemether injection
standard testing procedure Carbocisteine syrup
standard testing procedure Phytomenadione injection
standard testing procedure serratiopeptidase
standard testing procedure starch IP
standard testing procedure sucrose refined sugar
standard testing procedure titanium dioxide
standard testing procedure tramadol hydrochloride
standard testing procedure zinc sulphate
standard testing procedure croscarmellose sodium
standard testing procedure colour erythrosine supra
standard testing procedure magnesium hydroxide
standard testing procedure diclofenac sodium
standard testing procedure dibasic calcium phosphate
standard testing procedure cyanocobalamin
standard testing procedure cholecalciferol
standard testing procedure Calcium carbonate oyster shell powder
standard test procedure Calcium Citrate
standard testing procedure Bronopol
standard testing procedure Bromhexine Hydrochloride
Standard Testing Procedure diclofenac sodium injection
Standard Testing Procedure Drotaverine Hydrochloride injection
Standard Testing Procedure Tranexamic acid injection
standard test procedure paracetamol infusion
standard test procedure ofloxacin and ornidazole infusion
standard test procedure ornidazole injection
standard test procedure Ondansetron injection
standard test procedure dextrose injection
standard test procedure ciprofloxacin injection
STP and analysis method of Ammonium Chloride
analysis method of Losartan Potassium and Hydrochlorothiazide
analysis method of Linezolid Dry Syrup
analysis method of Drotaverine Hydrochloride and Mefenamic acid
Analysis method of Ceftriaxone Sodium and Sulbactam sodium Injection
analysis method of Cefepime and Tazobactam Injection
Analysis method of Hydroquinone Cream
Analysis method of Tacrolimus Ointment
Analysis method of Terbinafine HCL Cream
Analysis method of Mometasone Furoate and Fusidic Acid Cream
Analysis method of Disodium Hydrogen Citrate Syrup
Analysis method of Hydroquinone with Tretinoin Cream
Analysis method of Hydroquinone Tretinoin and Mometasone Furoate Cream
Analysis method of Sertaconazole Nitrate Cream
Analysis method of Halobetasol Propionate Cream
Analysis method of Povidone Iodine with Ornidazole Ointment
Analysis method of Eberconazole Cream
Analysis method of Luliconazole Cream
Analysis method of Fluconazole Gel
Analysis method of Ketoconazole Cream
Analysis method of Salbutamol and Choline theophyllinate Syrup
Analysis method of Methylcobalamin Injection
Analysis method of Piroxicam and paracetamol Injection
Analysis method of Alpha Beta Arteether Injection
Analysis method of Enrofloxacin Suspension