standard testing procedure Calcium carbonate oyster shell powder
Storage Requirements:
Store protected from light and moisture.
Sampling:
Sample from each container / bag and collect a minimum of 5g from each of the randomely selected containers/bags into indiviually no toxic,
selfsealing transparent polyethylene bearing ‘Sample For Analysis’ label kept in another transperent self sealing polythene bag.
Afetr completion of sampling return rest sample on the same container. Collect control sample in Pet bottle/Glass bottle.
Quantity of Composite Sample:
15 g
Quantity of Control Samle:
2 X 15 g
Description: White, crystalline or microcrystalline powder.
Solubility: Practically insoluble in water and in ethanol (95%), slightly soluble in water containing
carbon dioxide or ammonium salt; It is soluble with effervescence in dilute acids.
Identification:
A. By Chemical: 0.2 ml of the filtrate (Solution A) gives reactions A and B of calcium salts
B. By Chemical: Gives reaction A of carbonates.
Substances insoluble in acetic acid: The residue weighs NMT 10mg (0.2 per cent).
Arsenic: The resulting solution complies with the limit test for arsenic (4ppm).
Heavy Metals: NMT 20ppm
Barium: The solution remains clear for not less than 15 minutes.
Iron: NMT 200ppm
Magnesium & alkali metals: The residue weighs not more than 5 mg (1.0 per cent).
Chlorides: NMT 250ppm
Sulphates: NMT 0.3 %
Loss on Drying: NMT 2.0%w/w
Assay: 98.0% to 100.5%w/w on the dried basis.
Description: White, crystalline or microcrystalline powder.
Reporting: Report as Complies/Does not comply.
Solubility: Use the following quantities:
It is soluble with effervescence in dilute acids : 1 g in 10 to 30ml.
Slightly insoluble in water containing carbon dioxide or ammonium salt : 0.1 g in 10 to 100ml .
Practically insoluble in water and ethanol (95%) : 0.01 g in 100ml or more.
Reporting: Report as Complies/Does not comply.
Identification:
(A) Dissolve 5.0g in 80 ml of 2M acetic acid when effervescence ceases, boil the solution for 1 minute,
allow to cool, dilute to 100 ml with 2M acetic acid and filter 0.2 ml of the filtrate (Solution A) gives
reactions a and b of calcium salts.
(a) To 0.2 ml of the add filtrate add 0.5 ml potassium ferrocynide solution, the solution remains
clear. Add about 50 mg of ammonium chloride, a white crystalline precipitate is formed.
(b) To 0.2 ml of the filtrate, add 0.2 ml of a 2% w/v solution of ammonium oxalate. A white
precipitate is obtained that is only sparingly soluble in dilute acetic acid but is soluble in HCl
Reporting: Report as Complies/Does not comply.
(B) Gives reaction A of Carbonates:
Suspend 0.1g of the substance in a test tube in 2ml water. Add 2ml of 2M acetic acid. Close the tube
Immediately using a stopper fitted with a glass tube bent at two right angles Heat gently and collect
the gas in 5ml of 0.1 M barium hydroxide. A white precipitate us formed
Reporting: Report as Complies/Does not comply.
Substances insoluble in acetic acid:
Wash any residue obtained in Identification test A with four quantities, each of 5 ml of hot water and dry at 100° for 1 hour;
the residue weighs not more than 10 mg (0.2 per cent).
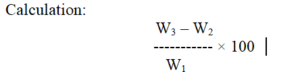
Where: W1 = Weight of sample, W2 = Weight of sintered glass & W3 = Weight of sintered glass with residue.
Reporting: Report as Value.
Arsenic:
Dissolve 2.5g in15 ml brominated hydrochloric acid and 45ml of water and remove the excess of
bromine with a few drops of stannous chloride solution AST. The resulting solution complies with
the limit test for arsenic.
Reporting: Report as Complies/Does not comply.
Heavy Metals:
To 1g add 5 ml water, add 8 ml of dilute hydrochloric acid, the latter being added slowly, shake and
evaporate to dryness on a water-bath. Dissolve the residue in 20ml of water, filter, add to the filtrate
3 ml of dilute acetic acid and water to make 25ml. This solution complies for the limit test.
Reporting: Report as Complies/Does not comply.
Barium:
Dissolve 0.6 g in 10 ml of 2M acetic acid by boiling cool and add 10 ml of calcium sulphate solution,
the solution remains clear for not less than 15 minutes.
Reporting: Report as Complies/Does not comply.
Iron:
Dissolve 0.2 g in 5 ml water and 0.5 ml of iron-free hydrochloric acid. Boil and dilute to 40ml with water
the solution complies with the limit test for iron.
Magnesium & alkali metals:
Dissolve 1.0g in10 ml of dilute hydrochloric acid, neutralise the solution by adding dilute ammonia
solution. Heat the solution to boiling and add 50ml of hot ammonium oxalate solution. Cool, dilute to
100 ml with water & filter.
To 50ml of the filtrate, add 1.5 ml of dilute sulphuric acid evaporate to dryness on a water-bath, heat the residue
to redness, allow to cool and weigh. The residue weighs not more than 10 mg.
Reporting: Report as Value.
Chloride:
1g dissolved in water by the addition of 3 ml of nitric acid complies with the limit test for chlorides.
Reporting: Report as Complies/Does not comply.
Sulphates:
Suspend 50.0mg in 5ml of water and add drop wise sufficient dilute hydrochloric acid to effect solution. Add 2ml of
dilute hydrochloric acid; the resulting solution complies with the limit test for sulphates.
Reporting: Report as Value.
Loss on Drying:
Determined on 1.0 g by drying in an oven at 200°C till constant weight.

Reporting: Report in %.
Assay:
Weigh accurately about 0.1g and dissolve in 3ml of dilute hydrochloric acid and 10ml of water. Boil for
10 minutes, cool and dilute to 50ml with water. Titrate with 0.05M disodium edetate to within a few ml of the expected end-point, add 8 ml of
sodium hydroxide solution and 0.1g of calcon mixture and continue the titration until the colour of the solution changes from pink to a full blue colour.
1 ml of 0.05M disodium edetate is equivalent to 0.005004g of CaCO3.

Analysis of vitamin B1 B6 B2 Nicotinamide and sodium pentothenate Injection
Analysis for Nandrolone Decanoate injection
Analysis of Dicyclomine and Diclofenac sodium Injection
standard testing procedure of Fexofenadine and Phenylephrine suspension
standard testing procedure of Piroxicam Injection
STP of Fungal Diastase and Papain capsules
standard testing procedure PVC
standard testing procedure glass ampoule
Standard testing procedure of Iron Sucrose Injection
Standard testing procedure lactose
Standard testing procedure mefenamic acid
standard testing procedure domperidone
standard testing procedure flavour mixed fruit
standard testing procedure dicyclomine hydrochloride
Standard testing procedure honey pure
standard testing procedure dextromethorphan hydrobromide
standard procedure of levocarnitine injection
Analysis of Ivermectin Suspension
standard testing procedure artemether injection
standard testing procedure artemether injection
standard testing procedure Carbocisteine syrup
standard testing procedure Phytomenadione injection
standard testing procedure serratiopeptidase
standard testing procedure starch IP
standard testing procedure sucrose refined sugar
standard testing procedure titanium dioxide
standard testing procedure tramadol hydrochloride
standard testing procedure zinc sulphate
standard testing procedure croscarmellose sodium
standard testing procedure colour erythrosine supra
standard testing procedure magnesium hydroxide
standard testing procedure diclofenac sodium
standard testing procedure dibasic calcium phosphate
standard testing procedure cyanocobalamin
standard testing procedure cholecalciferol
standard testing procedure Calcium carbonate oyster shell powder
standard test procedure Calcium Citrate
standard testing procedure Bronopol
standard testing procedure Bromhexine Hydrochloride
Standard Testing Procedure diclofenac sodium injection
Standard Testing Procedure Drotaverine Hydrochloride injection
Standard Testing Procedure Tranexamic acid injection
standard test procedure paracetamol infusion
standard test procedure ofloxacin and ornidazole infusion
standard test procedure ornidazole injection
standard test procedure Ondansetron injection
standard test procedure dextrose injection
standard test procedure ciprofloxacin injection
STP and analysis method of Ammonium Chloride
Analysis method of aceclofenac
analysis method of Losartan Potassium and Hydrochlorothiazide
analysis method of Linezolid Dry Syrup
analysis method of Drotaverine Hydrochloride and Mefenamic acid
Analysis method of Ceftriaxone Sodium and Sulbactam sodium Injection
analysis method of Cefepime and Tazobactam Injection
Analysis method of Hydroquinone Cream
Analysis method of Tacrolimus Ointment
Analysis method of Terbinafine HCL Cream
Analysis method of Mometasone Furoate and Fusidic Acid Cream
Analysis method of Disodium Hydrogen Citrate Syrup
Analysis method of Hydroquinone with Tretinoin Cream
Analysis method of Hydroquinone Tretinoin and Mometasone Furoate Cream
Analysis method of Sertaconazole Nitrate Cream
Analysis method of Halobetasol Propionate Cream
Analysis method of Povidone Iodine with Ornidazole Ointment
Analysis method of Eberconazole Cream
Analysis method of Luliconazole Cream
Analysis method of Fluconazole Gel
Analysis method of Ketoconazole Cream
Analysis method of Salbutamol and Choline theophyllinate Syrup
Analysis method of Methylcobalamin Injection
Analysis method of Piroxicam and paracetamol Injection
Analysis method of Alpha Beta Arteether Injection
Analysis method of Enrofloxacin Suspension
Analysis method of Levetiracetam Syrup
Analysis method of Sucralfate suspension
Analysis method of Sucralfate and Oxetacaine Suspension
Analysis method of Quinine Sulphate Suspension
Analysis method of Calcium Carbonate vitamin D3 Zinc Gluconate and Magnesium hydroxide suspension
Analysis method of Suspension of Tribasic Calcium phosphate with vitamin D3 and Vitamin B12
Analysis method of Calcitriol with calcium citrate Suspension
Analysis method of Oxyclozanide and Fenbendazole Suspension
Analysis method of Oxyclozanide and Levamisole Suspension
Analysis method of Triclabendazole and Ivermectin Suspension
Analysis method of Itraconazole Solution
Analysis method of Levocetirizine Dihydrochloride syrup
Analysis method of Iron Calcium Vitamin D3 Folic Acid Vitamin B12 Suspension
Analysis method of Ferrous Ascorbate Cyanocobalamin and Folic Acid Suspension
Analysis method of Ambroxol Hydrochloride Drops
Analysis method of Ferrous Ascorbate with Folic Acid suspension
Analysis method of Piracetam Syrup
Analysis method of Rafoxanide and Levamisole suspension
Analysis method of Zinc gluconate Syrup
Analysis method of Magaldrate Simethicone and Oxetacaine suspension
Analysis method of mefenamic acid and paracetamol suspension
Analysis method of Cholecalciferol Drops
Analysis method of Racecadotril suspension
Analysis method of Deflazacort Suspension
Analysis method of Montelukast sodium and levocetirizine Dihydrochloride Syrup
Analysis method of Iron and Folic Acid Syrup
Analysis method of Cyproheptadine Hydrochloride and Tricholine Citrate Syrup
Analysis method of Levofloxacin Hemihydrate Ornidazole and Vitamin E Solution
Analysis method of Albendazole and ivermectin in oral liquid