Analysis method of Levocetirizine Dihydrochloride syrup
1.0 OBJECTIVE
1.1 To lay down a procedure for analysis of Levocetirizine Dihydrochloride Syrup.
2.0 SCOPE
2.1 This procedure is applicable to the analysis of Levocetirizine Dihydrochloride Syrup quality control laboratory
3.0 RESPONSIBILITY
3.1 Q.C- Chemist
4.0 ACCOUNTABILITY
4.1 Manager-Quality Assurance
5.0 PROCEDURE
5.1 Description: Pour 50ml finish sample in beaker and observed visually.
5.2 pH: Taken 50 ml sample in beaker rinse the pH electrode first with purified water followed by sample dip the electrode in sample and observed the pH.
5.3 Volume variation: Measured the volume by measuring cylinder and determine the volume variation.
5.4 Identification:
5.4.1 In the assay, the principle peak in the chromatogram obtain with the test solution correspond to the peak in the chromatogram obtained with the reference solution.
5.5 ASSAY:
Each 5.0 ml contains:
Levocetirizine Dihydrochloride I.P. 2.5 mg.
Estimation Levocetirizine Dihydrochloride:
Method of Levocetirizine Dihydrochloride:
By HPLC:
BUFFER: Dissolve 3.85 Ammonium Acetate 1000 ml of distilled water adjust to ph. 5.5 with Glacial Acetic acid.
MOBILE PHASE: BUFFER : METHANOL
20 : 80
Standard preparation: Weight accurately about 50mg std wt. of Levocetirizine Dihydrochloride diluted to 100 ml volumetric flask with mobile phase. Further dilution 5.0 to 50 ml with mobile phase
Sample preparation: Taken 10ml sample diluted to 100 ml volumetric flask with mobile phase.
Chromatographic condition:
Wavelength – 220nm.
Flow rate – 1.2 ml/mint.
Injection volume -50 µl.
Column – C18 125mm x4.6 (5.0 µm.)
Temperature – 250c
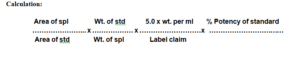
Calculation:
6.0 ABBREVIATIONS
| Sr. No. | Abbreviation used | Full form of abbreviation used |
| 1.0 | STP | Standard Testing Procedure |
| 2.0 | QA | Quality assurance |
| 3.0 | STD | Standard |
| 4.0 | SPL | Sample |
| 5.0 | I.P. | Indian Pharmacopeia |
| 6.0 | NM | Nano meter |
7.0 (ANNEXURE)
NA
8.0 REFERENCE
| Sr. No. | Reference Title |
| 01 | In House |
Bacterial endotoxin test for dexamethasone injection
Analysis of vitamin B1 B6 B2 Nicotinamide and sodium pentothenate Injection
Analysis for Nandrolone Decanoate injection
Analysis of Dicyclomine and Diclofenac sodium Injection
standard testing procedure of Fexofenadine and Phenylephrine suspension
standard testing procedure of Piroxicam Injection
STP of Fungal Diastase and Papain capsules
standard testing procedure PVC
standard testing procedure glass ampoule
Standard testing procedure of Iron Sucrose Injection
Standard testing procedure lactose
Standard testing procedure mefenamic acid
standard testing procedure domperidone
standard testing procedure flavour mixed fruit
standard testing procedure dicyclomine hydrochloride
Standard testing procedure honey pure
standard testing procedure dextromethorphan hydrobromide
standard procedure of levocarnitine injection
Analysis of Ivermectin Suspension
standard testing procedure artemether injection
standard testing procedure artemether injection
standard testing procedure Carbocisteine syrup
standard testing procedure Phytomenadione injection
standard testing procedure serratiopeptidase
standard testing procedure starch IP
standard testing procedure sucrose refined sugar
standard testing procedure titanium dioxide
standard testing procedure tramadol hydrochloride
standard testing procedure zinc sulphate
standard testing procedure croscarmellose sodium
standard testing procedure colour erythrosine supra
standard testing procedure magnesium hydroxide
standard testing procedure diclofenac sodium
standard testing procedure dibasic calcium phosphate
standard testing procedure cyanocobalamin
standard testing procedure cholecalciferol
standard testing procedure Calcium carbonate oyster shell powder
standard test procedure Calcium Citrate
standard testing procedure Bronopol
standard testing procedure Bromhexine Hydrochloride
Standard Testing Procedure diclofenac sodium injection
Standard Testing Procedure Drotaverine Hydrochloride injection
Standard Testing Procedure Tranexamic acid injection
standard test procedure paracetamol infusion
standard test procedure ofloxacin and ornidazole infusion
standard test procedure ornidazole injection
standard test procedure Ondansetron injection
standard test procedure dextrose injection
standard test procedure ciprofloxacin injection
STP and analysis method of Ammonium Chloride
Analysis method of aceclofenac
analysis method of Losartan Potassium and Hydrochlorothiazide
analysis method of Linezolid Dry Syrup
analysis method of Drotaverine Hydrochloride and Mefenamic acid
Analysis method of Ceftriaxone Sodium and Sulbactam sodium Injection
analysis method of Cefepime and Tazobactam Injection
Analysis method of Hydroquinone Cream
Analysis method of Tacrolimus Ointment
Analysis method of Terbinafine HCL Cream
Analysis method of Mometasone Furoate and Fusidic Acid Cream
Analysis method of Disodium Hydrogen Citrate Syrup
Analysis method of Hydroquinone with Tretinoin Cream
Analysis method of Hydroquinone Tretinoin and Mometasone Furoate Cream
Analysis method of Sertaconazole Nitrate Cream
Analysis method of Halobetasol Propionate Cream
Analysis method of Povidone Iodine with Ornidazole Ointment
Analysis method of Eberconazole Cream
Analysis method of Luliconazole Cream
Analysis method of Fluconazole Gel
Analysis method of Ketoconazole Cream
Analysis method of Salbutamol and Choline theophyllinate Syrup
Analysis method of Methylcobalamin Injection
Analysis method of Piroxicam and paracetamol Injection
Analysis method of Alpha Beta Arteether Injection
Analysis method of Enrofloxacin Suspension
Analysis method of Levetiracetam Syrup
Analysis method of Sucralfate suspension
Analysis method of Sucralfate and Oxetacaine Suspension
Analysis method of Quinine Sulphate Suspension
Analysis method of Calcium Carbonate vitamin D3 Zinc Gluconate and Magnesium hydroxide suspension
Analysis method of Suspension of Tribasic Calcium phosphate with vitamin D3 and Vitamin B12
Analysis method of Calcitriol with calcium citrate Suspension
Analysis method of Oxyclozanide and Fenbendazole Suspension
Analysis method of Oxyclozanide and Levamisole Suspension
Analysis method of Triclabendazole and Ivermectin Suspension
Analysis method of Itraconazole Solution
Analysis method of Levocetirizine Dihydrochloride syrup
Analysis method of Iron Calcium Vitamin D3 Folic Acid Vitamin B12 Suspension
Analysis method of Ferrous Ascorbate Cyanocobalamin and Folic Acid Suspension
Analysis method of Ambroxol Hydrochloride Drops
Analysis method of Ferrous Ascorbate with Folic Acid suspension
Analysis method of Piracetam Syrup
Analysis method of Rafoxanide and Levamisole suspension
Analysis method of Zinc gluconate Syrup
Analysis method of Magaldrate Simethicone and Oxetacaine suspension
Analysis method of mefenamic acid and paracetamol suspension
Analysis method of Cholecalciferol Drops
Analysis method of Racecadotril suspension
Analysis method of Deflazacort Suspension
Analysis method of Montelukast sodium and levocetirizine Dihydrochloride Syrup
Analysis method of Iron and Folic Acid Syrup
Analysis method of Cyproheptadine Hydrochloride and Tricholine Citrate Syrup
Analysis method of Levofloxacin Hemihydrate Ornidazole and Vitamin E Solution
Analysis method of Albendazole and ivermectin in oral liquid